
- support
- info@evidentic.com
- +49 (0) 30 959 99 8831
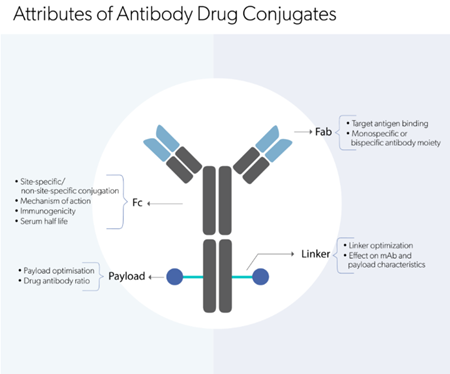
The demand for biotherapeutic drugs is on the rise. The possibilities of antibody and protein engineering have enabled the development of novel biologic drug formats with high specificity and low toxicity. Monoclonal antibody (mAb) drugs have shown remarkable success rates, and have paved way for the revolution of more innovative antibody-based drug modalities, including Antibody-drug conjugates (ADCs), bispecifics, antibody-fragments, single-domain antibody variants, chimeric antigen receptor T cells (CAR-Ts) and T cell receptors (TCRs).
The success of mAb therapy mainly in the treatment of cancer, autoimmune disorders and infectious diseases is achieved through the intrinsic properties of the mAbs:
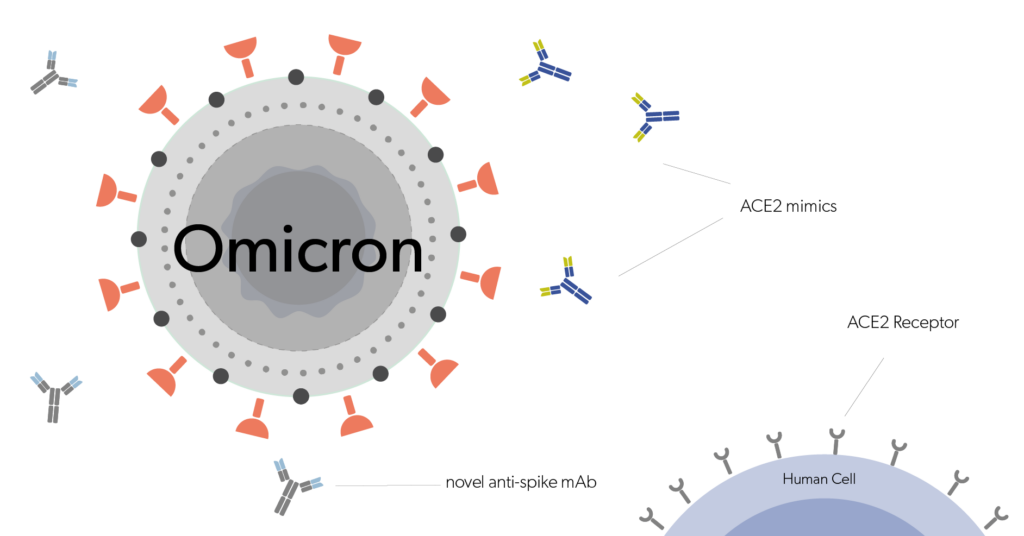
These properties pave the way for (1) high specificity, (2) enhanced safety and lower toxicity, (3) long half-life that allows infrequent dosing, and (4) better immune response modulating benefits to develop novel immunotherapies over the currently used small molecule drugs. Furthermore, in the treatment of viral diseases such as HIV or Hepatitis C, where a certain treatment or medicine is not in existence, mAb therapy proves useful as they act as neutralizing agents (by blocking cell-virus interactions) and/or promote the clearance of viral particles from the patient’s system and ameliorate patient survival and well-being.
In the recent decade, the pharmaceutical industry shifted its focus to expand its therapy portfolio by including and developing therapeutic antibodies. The advantages mentioned above of mAbs and the major technological advances have contributed to the rapid and efficient discovery and development of mAb-based therapies. Currently, across the globe, at least 600 therapeutic mAbs have been studied in clinical trials by commercial companies, and recently the 100th therapeutic mAb was approved by the United States Food and Drug Administration (US FDA), and all are currently on the market.
Buy clinical-grade molecules for your R&D projects from Evidentic.
Ab therapeutic discovery pipeline is categorized into phases, namely
Whereas target research to lead generation can be categorized under the early drug development phase.


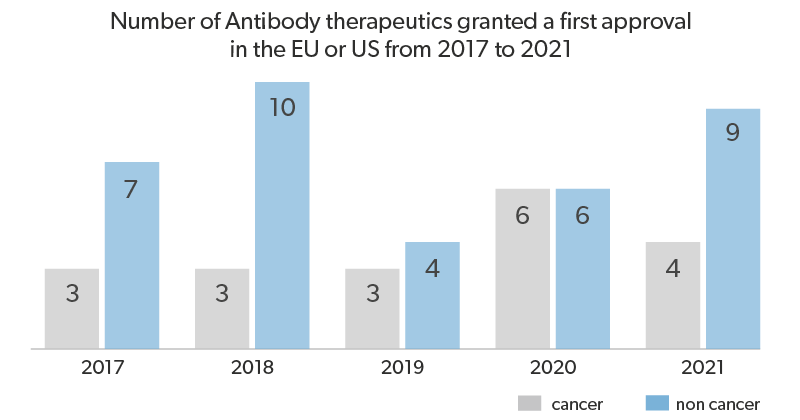

Evidentic GmbH
Martin-Buber-Str. 10
14163 Berlin