- support
- info@evidentic.com
- +49 (0) 30 959 99 8831
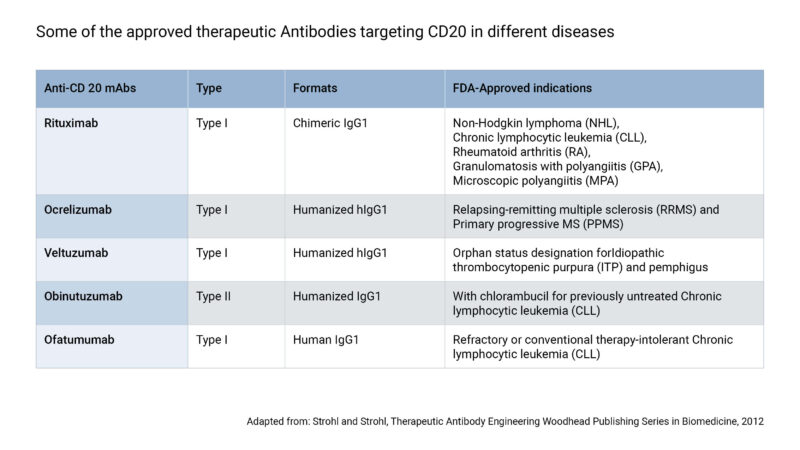
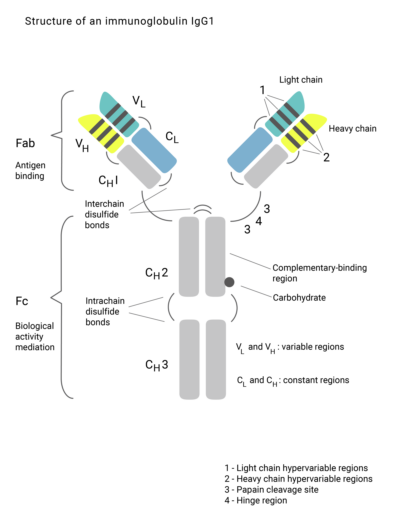
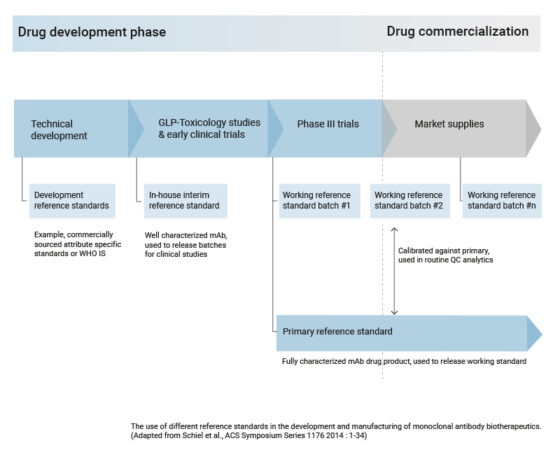
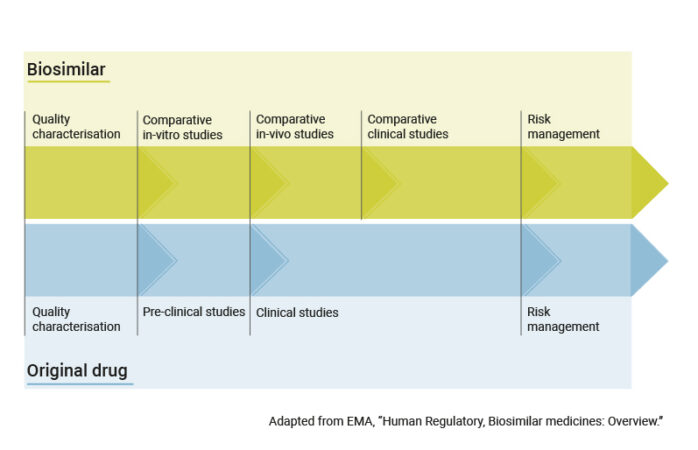
Here you can find information about biosimilars, from how they are produced to aspects of new developments regarding the research of these biologic drugs.



Evidentic GmbH
Martin-Buber-Str. 10
14163 Berlin