
- support
- info@evidentic.com
- +49 (0) 30 959 99 8831
Lack of treatment modalities for a disease or a clinical condition is the underlying reason for initiating a drug discovery programme. The establishment and undertaking of the drug discovery programme is an expensive affair. Therefore, it requires careful and detailed planning in any organization. In basic research, academicians often generate data to develop a hypothesis that the inhibition or activation of a protein or pathway will result in a therapeutic effect in a disease state. The hypothesis results in the target identification and selection leading to target validation before proceeding to the lead discovery phase, in which a drug-like small molecule or biological therapeutic is generated, screened and selected.
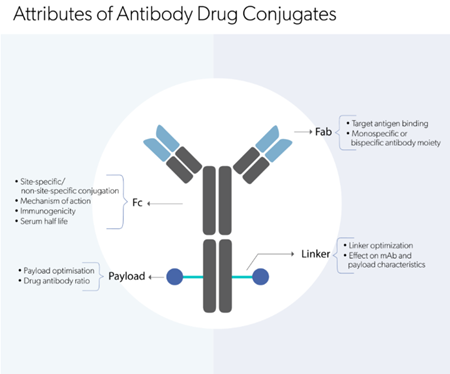
Target identification is one of the first critical steps in the drug development pipeline. An ideal target is described as a druggable target that is accessible to defined drugs such as a small molecule or biological molecule that, upon binding, evokes in vitro and in vivo measurable levels of biological and/or immunological response. Along with being druggable, other requisite characteristics of a target are being efficacious, safe, meeting the clinical and commercial needs. In the Antibody Drug Discovery process, target identification involves recognizing the molecular target of an Antibody, for example, the receptors or antigens expressed on tumor cells or other host cells. While choosing a target, factors such as target accessibility, expression and concentration should also be considered. Several approaches including:
are utilized in unison both individually or in combination for ideal target identification and selection. After identifying the target, a therapeutic strategy is conceived; in the case of antibody-based therapeutics, the three targeting strategies are an anti-ligand antibody, an anti-receptor antibody, and a receptor-Fc fusion protein.

Once the target has been identified, validation of the target is facilitated by obtaining relevant scientific evidence which exposes the target’s disease association and its therapeutic potential. The target validation can be conducted through various techniques ranging from in-vitro tools and whole animal models to modulation of the desired target in patients. Depending on the validity of each technique, any approach is chosen but a multi-validation approach provides increased confidence in the observed outcome. The different techniques include :
Furthermore, along with implementing target identification and validation techniques, key stakeholders’ approval is obtained together with establishing the project plan and forming the project team.
Emphasis is generally placed on the screening phase, but the impediment often arises in the early discovery phase when reagents and assays used in the screening preparation phase are not validated. Steps to be undertaken in the screening preparation phase to avoid downstream loss of time and waste of resources involves
Generally, reagent generation is routinely outsourced at many pharmaceutical companies to make drug discovery activities economical and cost-effective. Therefore, it is vital to evaluate the quality of the outsourced materials, for example, the degree of purity and aggregation, presence of endotoxin, rodent virus contamination, and bioactivity, prior to their application during the drug discovery process.
Evidentic provides approved clinical-grade molecules, as drug aliquots, for use as control reagents in early drug development. By using drug aliquots you will have:



Evidentic GmbH
Martin-Buber-Str. 10
14163 Berlin