
- support
- info@evidentic.com
- +49 (0) 30 959 99 8831
The revolutionary hybridoma technology, developed by Köhler and Milstein in 1975, made it possible to produce monoclonal antibodies (mAbs) in a large scale. This paved way for the translation of mAbs as a therapeutic modality with the first therapeutic mAb, the CD3-specific muromonab (OKT3), coming into existence in 1986. Therapeutic antibodies are now an important class of drugs that have applications in various therapeutic areas including cancer, inflammatory disease, organ transplantation, cardiovascular disease, pathogenic infection, respiratory disease and ophthalmological disease.
So far, nearly 79 mAb drugs have been approved for clinical use and more than 500 are in the clinical development pipeline. Therapeutic interventions using antibodies have been very successful, particularly in cancer and inflammatory diseases, which were previously considered as very difficult to treat.
Ever since the first mAb drug, antibody engineering technologies have drastically evolved. Each successive generation of mAb therapeutics was intended to enhance the clinical benefits and minimize toxicities. The main areas of focus in therapeutic mAb design are high target specificity, better systemic retention, and low immunogenicity. The following are the different generations of antibodies that have made an impact.
The murine antibodies were the first antibodies to be developed at a lab-scale using the hybridoma technology in rodents. But because of the differences between the human and rodent immune systems, usage of murine antibodies resulted in cytotoxicity. Thus their continuous administration often resulted in allergic reactions and anaphylactic shock, termed as human anti-mouse or anti-murine antibody (HAMA) response. Anti-CD3 mAb of murine origin (OKT-3), the first therapeutic mAb, approved for the treatment of transplantation rejection was discontinued primarily due to severe HAMA response in patients. Nevertheless, murine antibodies serve as frameworks for antibody development. Also due to the improved and optimized protocols for antibody generation from hybridoma mouse cell lines, this technology is considered popular and economical to produce new antibodies.
In order to minimize the HAMA response, chimeric antibodies were manufactured. These antibodies consist of 65% human genetic components by recombination of human constant regions and mouse variable regions in a suitable expression system. There are some chimeric antibodies approved by the FDA for use in human therapy and research, for eg., Infliximab, Rituximab, Abciximab. Presently even though there is a declining interest in developing chimeric antibodies for clinical applications, chimeric molecules can be widely used in the initial stage of antibody humanization strategies and also serve as controls and calibrators for specific research needs or in diagnostics applications.
The urgency to overcome the obstacles presented by low complementarity of the mouse line developed antibodies to the patient’s immune system, gave way to the creation of humanized antibodies. The humanized antibodies are close to 95% in human origin, with only the complementarity determining regions (CDRs) of the variable regions having mouse-sequence origin. However, these Abs sometimes present lower affinity than the parent murine mAbs with respect to binding with antigens. So as to increase the antibody-antigen binding affinity, techniques such as chain-shuffling randomization can be employed to introduce some transformations into the CDR. Daclizumab is the first FDA-approved humanized antibody, used in the treatment of multiple sclerosis.
Fully human sequence derived antibodies have no murine sequence and are largely produced via two sources: phage display technologies and transgenic mice. The first fully human sequence-derived antibody to be approved for therapeutic use was adalimumab (Humira), a fully human IgG1 antibody specific for TNFalpha that was selected via phage display of human VH and VL sequences.
The two main technology platforms that are currently used for manufacturing fully human mAbs are:
This method is used to produce diverse libraries of phage-displayed antibody variable regions (scFv or Fab) thus providing quick access of target-specific mAbs, without creating a library of hybrid clones. Through biopanning, an in-vitro screening technique using specific antigens, the phages expressing antibody fragments exhibiting better affinities are identified, sequenced and used for development of fully human mAbs.
The transchromosomal engineering process has aided the introduction of transgenes encrypting human immunoglobulin heavy and light chains, into mice germline. This has led to the successful development of mAb drugs from human-antibody producing mouse. Some of the advantages of this technology include direct generation of full-length IgG, more diversity and in-vivo affinity maturation. However, this method is not preferred for toxic antigens.

Technical advancement in biochemistry and antibody engineering has brought about new platforms for therapeutic antibody development. Some of these engineered Ab formats that are currently in clinical use are discussed below
BsAbs are artificially engineered mAbs that can bind to two epitopes. They can be a cost-effective alternative to combination therapy by targeting two different signaling pathways. The approach can also be used to effectively bind to a target cell and an immune cell to induce cytotoxic signaling. The currently approved BsAb formats are the full-length IgG-like asymmetric antibody and the single-chain variable fragment (scFv)-based Ab with no Fc fragment. Several other BsAb formats are under development and some in clinical evaluation phase.
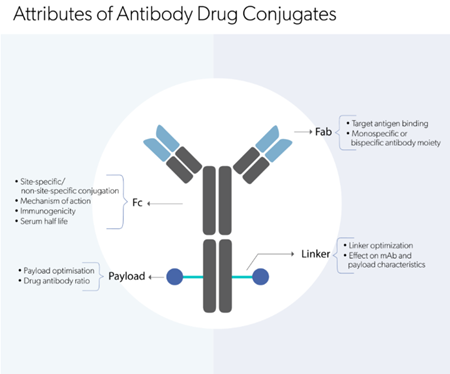
ADCs are an effective method to broaden the therapeutic window of primary Abs. The mAb can be conjugated to a cytotoxin drug or radioactive element via a non-cleavable linker. This approach increases therapeutic potency hence low doses can suffice the required clinical efficacy.
Therapeutic antibodies are usually homogenous preparations of monoclonal IgG1 monomers with identical protein sequences in order to ensure the desired biological effect in a consistent manner. In addition to specific receptor targeting and longer half-life in blood, the IgG class of antibodies exhibit effector functions that is now being tapped as a major therapeutic mode of action for clinical efficacy. Some of the key mechanisms of action (MoAs) exploited in antibody therapeutics are described here
The antibody can bind to the receptor on the target cell in an antagonistic fashion. The mAb can inhibit an undesired signaling pathway by blocking ligand-binding or interfering with the di/trimerization process to bring about the preferred physiological effect. For example, the mAb-ligand binding can give rise to inhibition of proliferative signals, blockade of immune checkpoints, induction of pro-apoptotic programs or re-sensitization of cell to a cytotoxic agent. Bispecific antibodies can target two ligands simultaneously so as to block redundant pathways.
Fc mediated response occurs when the target cell is opsonized with the several mAb drug molecules. The Fc region can bind to various molecules eliciting a cytotoxic response which can be via
(i) Antibody-dependent cell-mediated cytotoxicity (ADCC) – The Fc domain can bind to FcγRIIIA on NK cells triggering cell destruction via lytic factors secreted by the NK cells.
(ii) Complement‐dependent cytotoxicity (CDC) – The C1q subunit of the C1 compliment factor binds to the Fc domain initiating a signalling cascade that finally leads to the formation of a membrane attack complex (MAC), a pore that causes cell lysis.
(iii) Antibody-dependent cellular phagocytosis (ADCP or ADPh) – FcγRI expressed on macrophages, neutrophils, and eosinophils can bind to the Fc domain resulting in phagocytosis.
Here, the mAb-receptor binding activates a desired cellular pathway which is, in most cases, the activation of immune cells. Agonism of co-stimulatory ligands on T-cells and dendritic cells are currently being explored as an anti-cancer therapeutic strategy.
It is the main moA utilized by ADCs. Binding of the ADC to the target cell leads to endocytosis of the mAb complex, enabling intracellular delivery of the drug resulting in cell death. Certain payloads have the ability to diffuse and kill surrounding cells, a phenomenon termed as “bystander killing” which is useful in the case of solid tumors.
Most of the therapeutic antibodies elicit more than one of these pharmacological actions. Therefore, while developing mAb therapeutics, it is crucial to thoroughly evaluate the biological activity in-vitro and in in-vivo models using appropriate reference mAbs.
The current generations of antibody drugs demonstrate varied efficacy in patients. Advances in antibody engineering technology have enabled the development of novel mAb structures that are able to address the existing limitations and to enhance therapeutic potency. Amino-acid modifications and glycan engineering of the Fc domain have shown to improve the serum half-life and ADCC/CDC response. Antibody fragments are a new emerging class of therapeutic drugs that have lower immunogenicity and superior tumor penetration properties. Also, new formats of bispecifics such as tandem scFv’s and diabodies are being tested for their clinical efficacy. Last but not the least, intrabodies are a new addition to the bio-therapeutic armamentarium, consisting of antibodies that can target proteins within the cell.
Enhancing mAb therapeutic development with Evidentic!
With great progress in biotherapeutic drug discovery and antibody technology, the diversity in functions and targets for antibody therapy is ever-growing. Hence it is extremely important to assess the physical, chemical and biological properties of mAb molecules. Moreover, extensive studies must be conducted to understand their MoAs and downstream biological effects. Evidentic GmbH, a leading supplier of clinical-grade molecules as drug aliquots, provides approved biologics across various therapeutic areas, which can be used as a reference for comparative studies.
Discover clinical-grade molecules


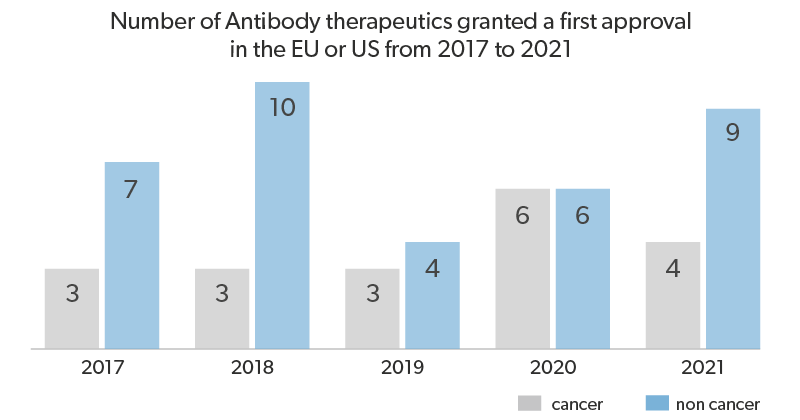

Evidentic GmbH
Martin-Buber-Str. 10
14163 Berlin