
- support
- info@evidentic.com
- +49 (0) 30 959 99 8831
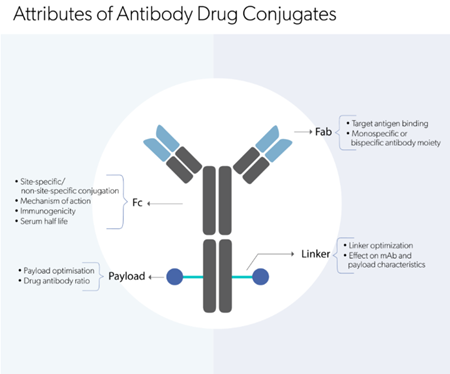
Once the target is validated, the type of intervention required is decided. Antibody drugs usually target cell surface molecules, for example, a cell-membrane receptor or a tumor antigen. Monoclonal antibody drugs can exert their therapeutic effect by various mechanisms of action, such as signal blockade, immune checkpoint inhibition or mAb internalization. Another important aspect to be considered at this point is the selection of an antibody format that can provide the desired therapeutic effect. Several parameters are taken into account, which includes the isotype (IgG, IgM or IgA), humanized vs fully human, monovalent vs multivalent, monospecific vs bispecific/multispecific, whole Ig vs Ab fragment, conjugate vs non-conjugate, etc.
The early antibody candidates are referred to as “hits”, produced by mAb clones generated using hybridoma technology, phage display or yeast display platforms. Therapeutic antibody candidates generated using the different technologies typically undergo the following R&D phases before becoming the final clinical candidate:
Successful screening of mAb clones requires high-quality reagents and well-validated functional assays. The common reagents employed for assay development are cDNA, expression plasmids, cell lines, purified proteins, control and reference antibodies, and target orthologs for testing species cross-reactivity. The screening assays typically include primary, secondary, and tertiary assays. Primary screening assays, generally ELISAs, are used to identify hits and filter out the antibodies that bind to the target of interest. Subsequently, secondary assays (high-throughput) and tertiary assays (low-throughput) are designed to assess the desired biological activity of candidate antibodies. For example, suppose the goal is to identify antibodies that block a particular ligand-receptor signalling pathway. In that case, a plate-based ligand/ receptor binding ELISA can be used as a secondary assay, and a cell-based ligand/receptor binding assay with a signalling readout can be employed as a tertiary assay.
In this phase, the selection of reference antibody, positive control antibody and negative control antibody is critical as they help make valid decisions. A suitable reference antibody or positive control antibody is an essential tool for assay validation. The reference antibody can be a commercially available monoclonal antibody with a function similar to the intended therapeutic candidate or an antibody reconstructed from sequences available in the public domain. Reference antibodies are also used for in-vivo proof-of-concept studies, either to validate the target or establish an efficacy model. Similarly, the negative control antibody to be used in assays is also very crucial. The negative control mAb used in cell-based functional assays and in-vivo efficacy studies must be species and isotype-matched.
Find relevant approved mAb drug aliquots for use as reference antibody and isotype controls at Evidentic!
Lead selection involves a rigorous, multi-step filtering process to select a lead molecule(s) that matches the pre-established criteria or critical quality attributes (CQAs), for advancement into the next phase of drug development. Firstly, the advanced hits undergo small-scale expression and purification for further evaluation and characterization. Antibody characterization includes structural, biochemical, biophysical and functional properties. For example, assays are designed to determine:
Read more: https://evidentic.com/characterization-of-therapeutic-monoclonal-antibodies/
At this stage, the “hits” that do not meet the CQA criteria are eliminated (For example, suboptimal target binding affinity, lack of ADCC function, or poor biochemical and/or biophysical attributes). After in-vitro characterization, the selected lead molecule(s) are expressed and purified for preliminary in-vivo efficacy testing. Crude PK/PD and toxicity studies can be conducted along with the efficacy studies in the relevant animal models to confirm the lead selection.
Benchmark your mAb candidates against Evidentic, approved drug aliquots!
Find relevant approved mAb drugs at Evidentic that have well-characterized structural, biophysical and functional properties for use as reference antibodies!

The selected lead molecule may require further molecular engineering to maximize the therapeutic efficacy and safety, minimize immunogenicity/toxicity, and improve manufacturability to become the final clinical candidate. The process includes:
The new generations of therapeutic antibodies are humanized or fully-human Ig sequences that possess low immunogenecities. This means they are more similar to the human Ig sequence and, therefore, induce low anti-drug antibody (ADA) reactions in the patient, which could affect the therapeutic efficacy. For example, in the case of rodent antibodies (produced by hybridoma technology), >90% of the IgG sequence is replaced with human IgG sequence. Computer-aided design, phage display and yeast display are the most widely used methods for antibody humanization. Some of the popular softwares used are BioLuminate, MOE and the webserver Tabhu (tools for antibody humanization)
Other factors such as epitope sequence, aggregation, dose, route of administration and target can also contribute to the immunogenicity of therapeutic antibodies. Deimmunization is the process of eliminating antigenicity epitopes, i.e. T cell, B cell and MHC epitopes, that can cause an immune reaction. Computational tools such as, Protean 3D from DNA star, are often used to predict these epitopes in the query sequences. Additionally, a novel approach to decrease immunogenicity is the introduction of Treg epitopes into the antibody structure that will stimulate Treg cells.
The process involves enhancing the ligand-binding affinity to the desired affinity range (normally 0.1–10 nM). For example, a high binding affinity is often necessary to block receptor signalling cascade. Moreover, affinity maturation helps reduce cross-reactivity to other related antigens, thereby decreasing the chances of off-target toxicities. Maturation technologies include random mutagenesis, targeted mutagenesis, chain shuffling and in-silico approaches (for example, BioLuminate).
The Fc region of antibodies can elicit immune effector functions such as antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-induced complement-dependent cytotoxicity (CDC), and antibody-dependent cell-mediated phagocytosis (ADCP). Fc engineering via in-silico designing, site-directed mutagenesis, and glycosyl modifications improve effector functions, especially in cancer therapeutics. On the contrary, antibody drugs for auto-immune diseases and inflammatory disorders require Fc mutations that disable Fc- FcγR interactions.
Pharmacokinetic improvement aims to increase serum half-life (allowing low dosage), increase dosing intervals, develop subcutaneous formulations, etc. Specific mutations in the Fc region have shown to reduce nonspecific clearance (when nonspecifically internalized into the cellular endosomes) and antigen binding-mediated internalization and clearance. For example, increasing Fc-FcRn binding at neutral pH allows antibody escape from degradation.
An ideal biologic drug product must possess high thermostability, high solubility, high chemical stability and low heterogeneity. These features collectively ensure successful retention of biological activity of the antibody during storage, minimal aggregation, higher production yields, ease of formulation and improved quality control in manufacturing.
Find aliquots of approved biologic commercial drugs at Evidentic as reference antibodies for lead optimization and characterization!
Discover clinical-grade molecules



Evidentic GmbH
Martin-Buber-Str. 10
14163 Berlin